
Feline Leukemia( FeLV) Ag rapid test kit


Feline Leukemia( FeLV) Ag rapid test kit
Principles
FIV Ab + FeLV Ag Combined Rapid Test is based on sandwich lateral flow immunochromatographic assay. The test device has two testing windows. Each testing window has an invisible T (test) zone and C (control) zone. When sample is applied into the sample hole on the device, the liquid will laterally flow on the surface of the test strip. If there is enough FIV antibody or FeLV antigen in the sample, a visible T band will appear in the corresponding testing window. The C band should always appear after a sample is applied, indicating a valid result. By this means, the device can accurately indicate the presence of FIV antibody or FeLV antigen in the sample.
For Cassette
1.Review “Specimen collection” instructions. Test device, patient’s samples, and controls should be brought to room temperature (20-30℃) prior to testing. Do not open pouches until ready to perform the assay.
2.Remove the test device from its protective pouch (bring the device to room temperature before opening the pouch to avoid condensation of moisture on the membrane). Label the device with patient or control number.
3.Hold the dropper vertically and transfer 3 full drops of urine (approx. 0.15ml) to the specimen well (S) of the test device, and then start the timer. Use a separate pipette and device for each sample or control.
4.Read result between 3 to 8 minutes after the addition of samples. Do not read result after 8 minutes.

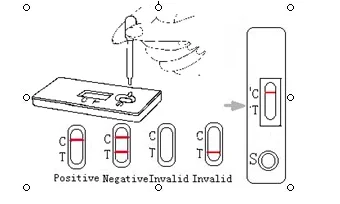
How To Read ?
Positive: Only one pink band appears on test region of the Cassette.
Negative: Two pink bands appear on test region of the Cassette.
Invalid: If without colored band appears on test region, this is an indication of a possible error in performing the test. The test should be repeated using a new device.



Feline Leukemia( FeLV) Ag rapid test kit
Principles
FIV Ab + FeLV Ag Combined Rapid Test is based on sandwich lateral flow immunochromatographic assay. The test device has two testing windows. Each testing window has an invisible T (test) zone and C (control) zone. When sample is applied into the sample hole on the device, the liquid will laterally flow on the surface of the test strip. If there is enough FIV antibody or FeLV antigen in the sample, a visible T band will appear in the corresponding testing window. The C band should always appear after a sample is applied, indicating a valid result. By this means, the device can accurately indicate the presence of FIV antibody or FeLV antigen in the sample.
For Cassette
1.Review “Specimen collection” instructions. Test device, patient’s samples, and controls should be brought to room temperature (20-30℃) prior to testing. Do not open pouches until ready to perform the assay.
2.Remove the test device from its protective pouch (bring the device to room temperature before opening the pouch to avoid condensation of moisture on the membrane). Label the device with patient or control number.
3.Hold the dropper vertically and transfer 3 full drops of urine (approx. 0.15ml) to the specimen well (S) of the test device, and then start the timer. Use a separate pipette and device for each sample or control.
4.Read result between 3 to 8 minutes after the addition of samples. Do not read result after 8 minutes.
How To Read ?
Positive: Only one pink band appears on test region of the Cassette.
Negative: Two pink bands appear on test region of the Cassette.
Invalid: If without colored band appears on test region, this is an indication of a possible error in performing the test. The test should be repeated using a new device.





- 步骤1通过搜索关键词找到您感兴趣的产品
- 步骤2请点击 "查询 "或 "请求会面 "按钮联系供应商,否则,您也可以咨询我们的进口顾问。
- 步骤3确认您的电子邮件后,得到供应商的回复,或我们的贸易经理会尽快与您联系。
- 步骤4与中国供应商建立无语言障碍的会议
 Testsealabs Rapid Test Dengue IgG/IgM test Kit Cheap price Variety complete Convenient
Testsealabs Rapid Test Dengue IgG/IgM test Kit Cheap price Variety complete Convenient




